
RHDV2 Resources
Locate a Vaccine
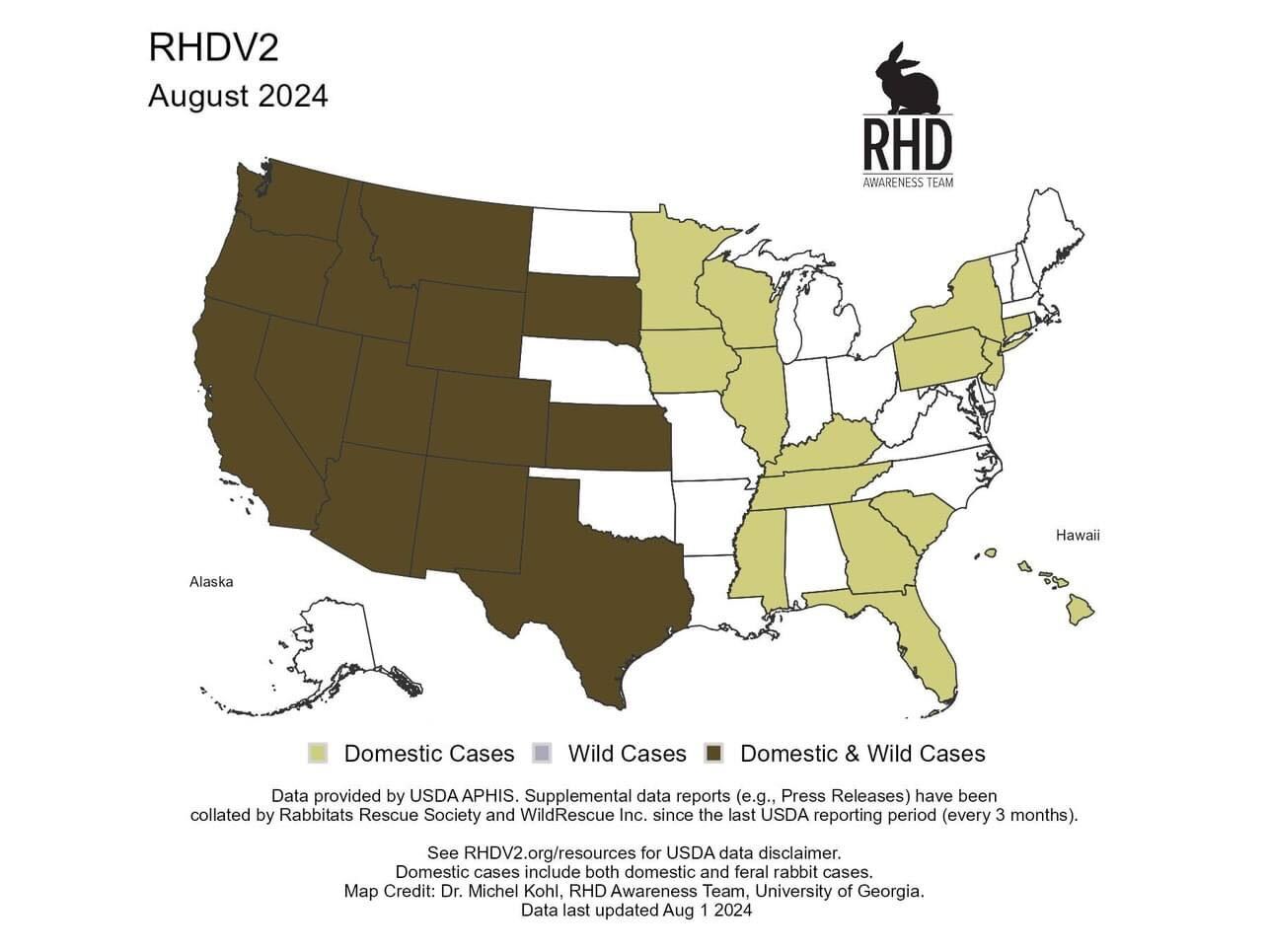
US Outbreak Status
(courtesy of rhdv2.org)
Rabbit Hemorrhagic Disease (RHDV2) is a highly contagious and lethal calicivirus with devastating consequences for both wild and domesticated rabbits. We strongly recommend vaccinating all rabbits, including those in shelters, with the safe and effective RHDV2 vaccine. If you board your rabbits, choose facilities that require vaccination. Report any sudden, suspicious deaths to your vet as potential RHD cases.
Symptoms
(courtesy of rabbitors.info)
- Peracute: animals will be found dead within a few hours of eating and behaving normally. This is most common. Many rabbits with the North American RHDV2 strain will not exhibit any external hemorrhaging (e.g. blood from nose).
- Acute: affected animals will show lethargy and a heightened fever (>40°C) with an increased respiratory rate, usually passing away within 12h.
- Subacute: rabbits will show mild or subclinical signs from which they recover and become immune to further RHDV.
Protecting Your Rabbit
Vaccinate
Vaccinate annually, especially in RHDV2-prone areas.
Indoors
Keep your house rabbits indoors, limit or eliminate outdoor playtime.
Avoid Playdates
Avoid letting rabbits play/interact with rabbits from outside your home.
Wash Hands
Wash your hands thoroughly before handling your rabbits.
Change clothes
Change clothes and wash hands after contact with rabbits from outside your household.
Avoid foraged foods
Don't feed locally foraged plants, hay grasses, or branches
No Shoes
Remove shoes and disinfect after outdoor activities near wild rabbit habitats, using RHDV-effective disinfectant. Consider a blanket "no shoes" policy in your home.
Minimize insect exposure
Minimize your rabbits' exposure to flying insects. Mosquitos and flies, in particular. Make sure your windows all have secure screens.
Quarantine
Quarantine new bunnies being added to your household for at least 14 days.
Annual Booster
The RHDV2 vaccine needs to be boosted annually to remain effective. Don't forget to get your rabbits boosted every year.
Further Reading
- RHDV2.org website
- Rabbit hemorrhagic disease article on WabbitWiki
- RHVD2 resources from The American Veterinary Medical Association (AVMA)
- RHD factsheet from the USDA
- Medgene RHDv2 FAQ (pdf)
- Medgene RHDv2 vaccine conditional use state approval list (pdf)
(current as of 16jan2024) - RHDV2 regulations in the US article on WabbitWiki
- Importing Pet Rabbits Between Canada and the USA
- Rabbit Hemorrhagic Disease News Network Facebook Group
- North Americans RHDV2 Group Facebook Group
See Also
- Myxomatosis
- Myxomatosis Case Reports
- Traveling With Your Rabbit
